Axis 1: The evolution of organic carbon and its impact on the production and evolution of secondary organic aerosol
As they oxidize, gaseous organic compounds contribute to the formation of tropospheric ozone, other photo-oxidizing species (such as free radicals), and secondary organic aerosol precursor compounds. Although many kinetic and mechanistic data are available for the reactivity of organic compounds in the gas phase, they remain insufficient to understand the reactivity of the wide variety of chemical structures present in the atmosphere, particularly multifunctional organic compounds formed after several oxidation steps of emitted precursors.
In order to improve our understanding of the evolutionary processes of organic compounds in the gas phase, our work aims to :
- determine the oxidation kinetics and mechanisms of gaseous organic compounds. These studies allow us to feed databases (EUROCHAMP for example) for oxidation kinetics that are still poorly documented, but also to identify the influence of the structure of organic molecules on their reactivity. To this end, we are running oxidation and photolysis experiments in the CSA and CESAM atmospheric simulation chambers.
- seek a link between the physico-chemical properties of organic molecules and their chemical structure. This work involves the development and/or evaluation of structure-property predictive methods to estimate missing data (kinetic constants, branching ratios, structure of products formed, saturation vapor pressure, Henry's law constant, etc.).
- evaluate our understanding of the reactivity of gaseous organic carbon. This work aims to identify missing and/or poorly constrained chemical processes. We are developing reference chemical mechanisms with the GECKO-A generator, evaluating these mechanisms by comparison with experiments in atmospheric simulation chambers, and exploring the evolution of gaseous organic matter.
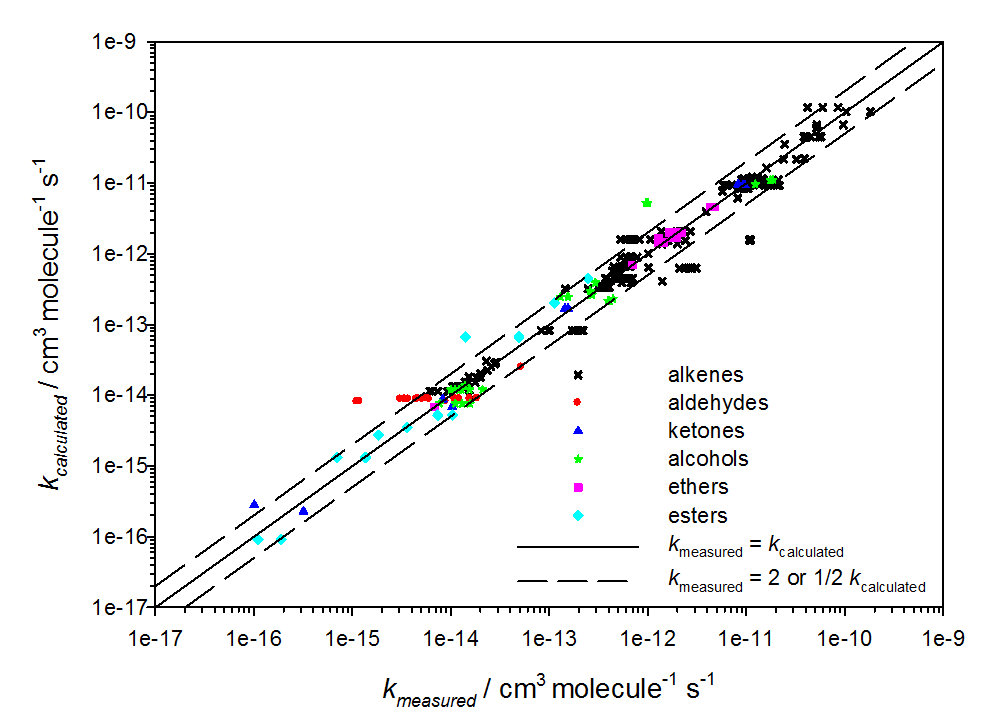
Evaluation de la relation de structure propriété développée pour estimer la réactivité des composés organiques avec NO3 (Kerdouci et al., 2014).

